DIY Cards

Shoreline Restoration
Event Card
Search your deck, your opponent’s deck, and/or either discard pile for a producer (level 1) card and put it into play. Reshuffle the deck(s).

Pacific Lugworm
Abarenicola pacifica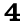


3 POINTS
Play: Abarenicola pacifica has a MOVE of 1.
Lugworms feed on detritus at the bottom of the river.

Buzzer Midge
Chironomus plumosus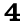


2 POINTS
Adult midges don’t feed. Midge larvae feed on suspended organic matter in water and mud.
Play: C. plumosus has a MOVE of 1.

Bullhead/Sculpin
Leptocottus armatus


6 POINTS
Play: Leptocottus armatus has a MOVE of 2.
Every spring it is celebrated in the Bullhead Derby at Gary Point Park.

Musqueam Fisheries
Event Card
Musqueam fisheries officers patrol the waters in their traditional territory, protecting against overfishing.
Play: If played IMMEDIATELY, this card counters the effect of OVERFISHING.

Overfishing
Event Card
Poor government regulation leads to overfishing. Roll a die. Remove the corresponding number of fish species from the game IMMEDIATELY.